Finding the way in
Editor's note: Ed Siebert is a principal of Integrated Marketing Associates, a Bryn Mawr, Pa., research firm.
Increasing competition in the managed care industry along with a need to service a growing number of national clients that require similar sets of benefits across their many national affiliates has caused a movement toward a more centralized business model. As the activities of managed care organizations (MCOs) become increasingly national in scope, smaller regional plans fall prey to larger organizations as the latter attempt to position themselves in a marketplace where corporate consolidation and increasing competition have become the norm. As many MCOs have acquired new regional affiliates, they have moved toward a national focus for their plans.
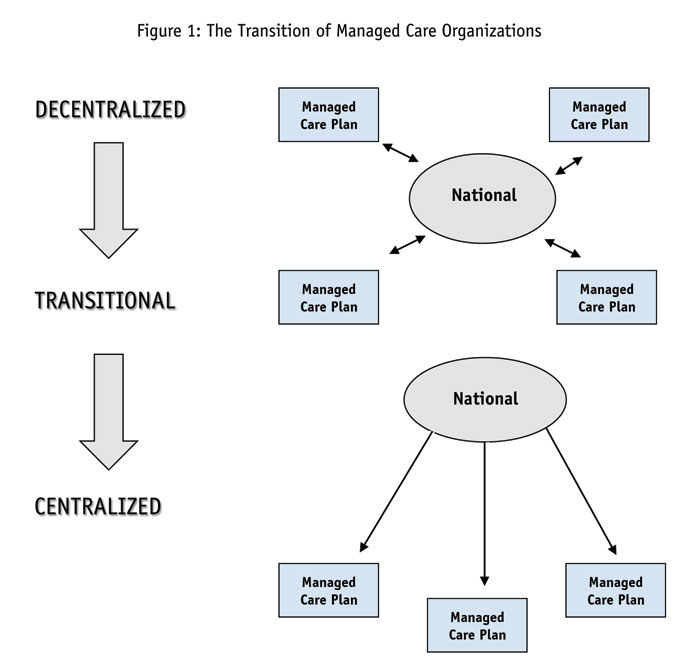
In examining the corporate structures of managed care organizations, it seems apparent that MCOs can be typified according to a set of criteria that would allow for the development of a typology of organizations. The typology laid out in Figure 1 and Table 1 identifies three general types of MCOs: the centralized, the transitional and the decentralized. Each of these categories is intended to place individual organizations within a cognitively-useful framework that should make classifying and understanding their organizational structures simpler.

Table 1: A Typology of Managed Care Organizations
Centralized
Structure: Typically characterized by top-down corporate management where regional plans have little or no input into corporate decision-making.
Formulary: Autocratic - tends to be generated through a strong national P&T committee with little input from regional or local plans.
Contracting: Either through a PBM or a process based at corporate headquarters.
DSM Programs: Tend to be lodged in the corporate headquarters and disseminated to regional/local plans.
Treatment Algorithms: Developed nationally and disseminated to regional/local plans.
Access Strategy: At the corporate level. These plans have procedural barriers in place to dissuade pharmaceutical companies from gaining access at the regional level. In some cases, persistent attempts to penetrate regional plans may hinder national relationships.
Transitional
Structure: Typically characterized by relatively strong corporate management but where regional/local plans have varying levels of autonomy.
Formulary: Quasi-democratic – tends to be driven by a national P&T committee but regional/local plans have input, sometimes significant.
Contracting: Usually carried out by a national PBM or by the national headquarters.
DSM Programs: May be developed either regionally or nationally.
Treatment Algorithms: Typically, national guidelines are established that regional/local plans may modify.
Access Strategy: At both regional and corporate levels, focus depends on program or business issue at hand. Move soon to develop a regional relationship since transitional plans are moving toward a more centralized structure, although these plans appear to be having some difficulty in attaining this centralization in the near-term.
Decentralized
Structure: Corporate organization serves role of consensual arbiter for regional plans. Slower response to corporate issues because rapid-acting leadership is lacking.
Formulary: Decentralized – national P&T is consensus-oriented with regional/local plans determining type and contents of formulary.
Contracting: Internally through national organization.
DSM Programs: Developed regionally.
Treatment Algorithms: Developed regionally but may lead toward national consensual guidelines.
Access Strategy: At both regional and corporate levels, with regional focus depending on goal of particular access. Typically contracting issues developed by corporate, while regional plans develop DSM and other programs.
Centralized
Centralized organizations are those that have consolidated their business practices into a strict hierarchical corporate structure, where the national headquarters maintains rigid control of nearly all functions of the organization. Regional plans serve to administer local benefits and address local market needs but they do so under the auspices of the national organization. Formulary design, contracting, disease state management (DSM) programs and treatment algorithms are all developed by the national organization and disseminated to the regional affiliates in a strict top-down manner. Local or regional plans may have some leeway to make modifications based on local market conditions but these tend to be minimal. While these kinds of organizations may be willing to work with pharmaceutical companies, access is clearly restricted to the corporate level. The best strategy would appear to be one that emphasizes cooperation with the corporate organization and deemphasizes attempts to access regional plans. In fact, some organizations may sever contact with a pharmaceutical company if the latter provided unauthorized information to regional organizations.
Transitional
Transitional MCOs are those that are moving toward consolidation of their business practices but have not yet reached a stage of strict centralization. Reasons appear to vary among organizations; however, it would seem that most MCOs falling into this category are still in the middle of their growth phase. They have recently acquired new regional plans and/or have only of late moved to a more national focus.
While it is likely that some of these MCOs may become more centralized as their new structure matures, it is also likely that some will remain less centralized since the degree of possible centralization may depend on the type of regional plans that are part of the corporate structure. Relatively homogenous organizations – those that are composed of the same type of plans, e.g., IPAs or PPOs – that do not require high degrees of regional autonomy will find centralization more natural.
On the other hand, those organizations composed of either more heterogeneous plans (those with multiple types of regional plans) or plans that require greater amounts of regional autonomy (such as group models) may find strict centralization more difficult, if not impossible, to achieve.
Transitional plans typically have centralized, yet quasi-democratic, formulary processes. With this type of structure, regional organizations tend to have some leeway over which medications are placed on formulary, either by direct vote on the national P&T or through other formal procedures. DSM programs may be developed either regionally or nationally. Treatment algorithms are usually developed as national guidelines but regional plans may have significant leeway in how those algorithms are applied. Pharmaceutical company access to regional organizations tends to be less restrictive within transitional MCO plans, although the degree of regional access appears dependent on the focus of entry. For example, MCOs in a transitional stage may clearly restrict the access of pharmaceutical companies to the national organization for formulary issues but allow broad access to regional organizations for the development of DSM programs.
Decentralized
As its name implies, the decentralized category of MCOs describes those organizations that tend to have a relatively weak centralized structure. In these cases, the corporate headquarters tends to serve more as an arbiter for the regional plans than as a directing, centralizing force. Guidelines for various corporate programs may be developed at the national level but it is apparent that key decision-making is carried out regionally. While the formulary is established by the national organization, regional organizations have defining authority. The decentralized MCO may be accessed through both regional and national levels; however, it provides the most open of all organizations since regional plans may be approached on an independent basis.
Implications for the pharmaceutical industry
As discussed earlier, the more centralized MCOs generally control the business practices of their regional affiliates in most areas, including in formulary, general disposition of benefits (such as hospitalization and emergency procedures), disease state management, treatment algorithms and other areas. Those MCOs classified as transitional in nature tend to allow their regional affiliates a greater degree of autonomy in some business areas such as local sales, marketing and credentialing, yet tend to retain control of formulary and other key benefit areas. Lastly, decentralized MCOs allow their regional plans the greatest degree of autonomy in areas such as establishing benefits, DSM and other programs and may in fact have little control over key areas of business, including the final form of the formulary.
The typology presented here would clearly be useful in the development of a general strategy for each type of organization. Such an approach has advantages over one that would simply profile individual organizations, since once a strategy has been developed for each category of organization, it need only be adjusted (through specific tactics) for the individual MCOs within that category. This approach should save pharmaceutical companies substantial time and resources over an approach that requires a considerable reinvestment for each organization. This approach also should limit the time and resources spent on attempting to gain access to MCOs at both the regional and corporate levels.
Research can be instrumental
Marketing research can be instrumental in helping pharmaceutical companies to determine the most effective methods of approaching each type of MCO. Individual interviews – most easily and anonymously conducted over the phone – can help identify the decision-making chains within key payer organizations in each territory. In particular, companies may gain a better understanding of the influence of regional offices regarding formulary decisions, contracting and development of treatment algorithms.
Discussions with payer decision makers may also provide direction with regard to the most advantageous approach routes for sales representatives, pointing them toward offices and departments that are most likely to be open to discussion and suggestions. Identification of the most-open pathways within each type of MCO should help provide opportunities for advancing rather than hindering relationships between manufacturer and payer.